 
Excited states are situation where one or more electrons within an atom are at a higher potential energy than they would be in the ground state. Second, when atoms are heated in a flame or energized with electricity First, theĮlectron can absorb a photon of just the right amount ofĮnergy to move it from one quantum shell to another. 
Sometimes they can be promoted to anĮlectron shell with a higher potential energy. Just as a ball sitting on the ground has its lowest potential energy, electrons which are close to the nucleus are similarly low in potential energy.Įlectrons do not always stay in the ground When they are there, they have their lowest possible potential energy. In this state electrons are as close to the atom’s nucleus as they are allowed to be. When all of the electrons are at the lowest possible energy The electrons in an atom occupy different energy levels. Ground states by emitting light of very specific colors. These colors comeįrom the excitation of electrons which then resume their 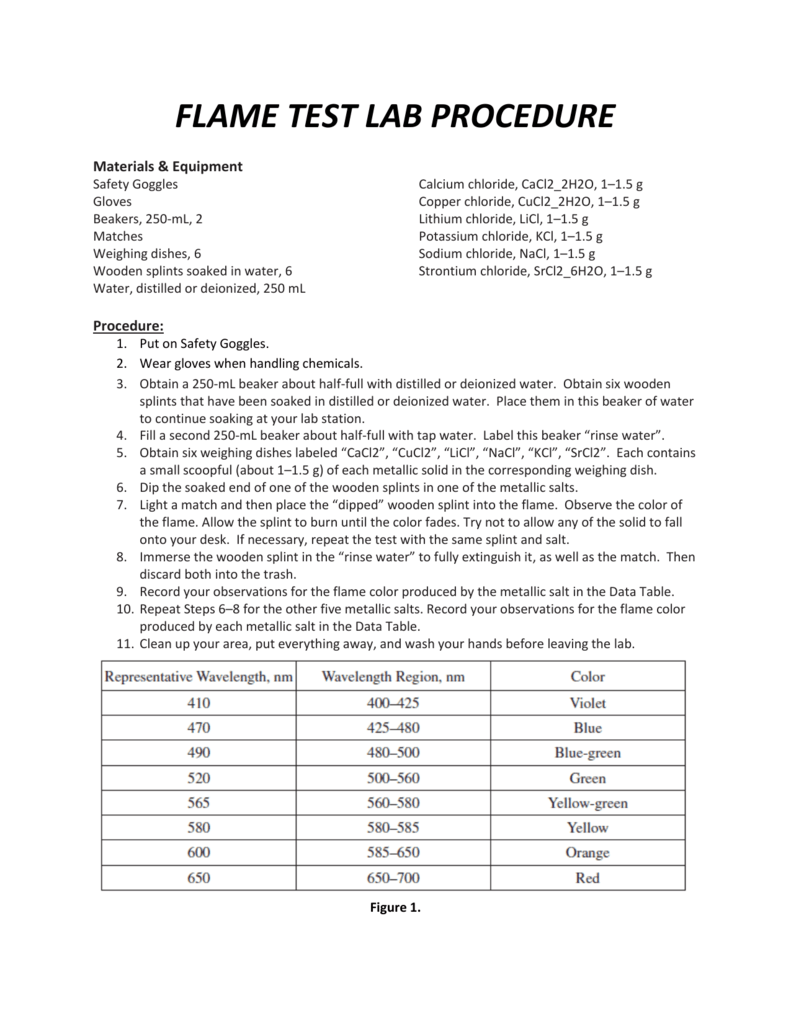
Students will observe small samples of chloride salts ofĭifferent metals. In this lab students will learn about atomic energy levels,Įmission spectroscopy, and flame tests for element It may also be viewed by students who were Lab activity may enhance the learning it is meant toĪlso, I have produced a video suitable as an introduction About fireworks (titled Fireworks!) before or after this 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
